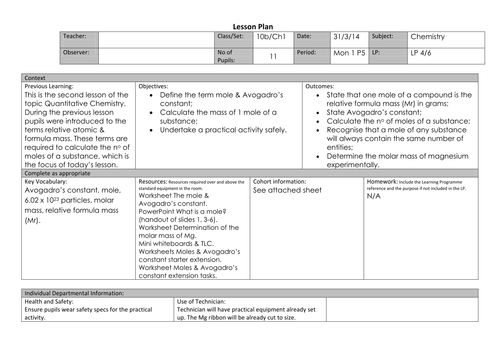
43 the mole and avogadro's number worksheet
Skills Worksheet Problem Solving - School of Arts & Sciences The number 6.022 137 1023 is called Avogadro’s number. For most purposes it is rounded off to 6.022 1023. Because this is an awkward number to write over and over again, chemists refer to it as a mole (abbreviated mol). 6.022 1023 objects is called a mole, just as you call 12 objects a dozen. Look again at how these quantities are related. 55.847 g of iron 6.022 1023 iron atoms … Class 9 Science CBSE worksheet for Atoms and Molecules Atoms and Molecules Worksheet. Given below are the Class 9 Science CBSE worksheet for Atoms and Molecules (a) Short questions (b) True and False problems (c) Fill in the blank's Hope you like them and do not forget to like , social share and comment at the end of the page. Fill In The Blanks. During a chemical reaction, the sum of the _____ of the reactants and products …
IF8766 Mole - Avogadro - Mass Grams - key.pdf How many moles are in the number of molecules below? ... One mole of a substance contains Avogadro's Number (6.02 x 1023) of molecules.2 pages

The mole and avogadro's number worksheet
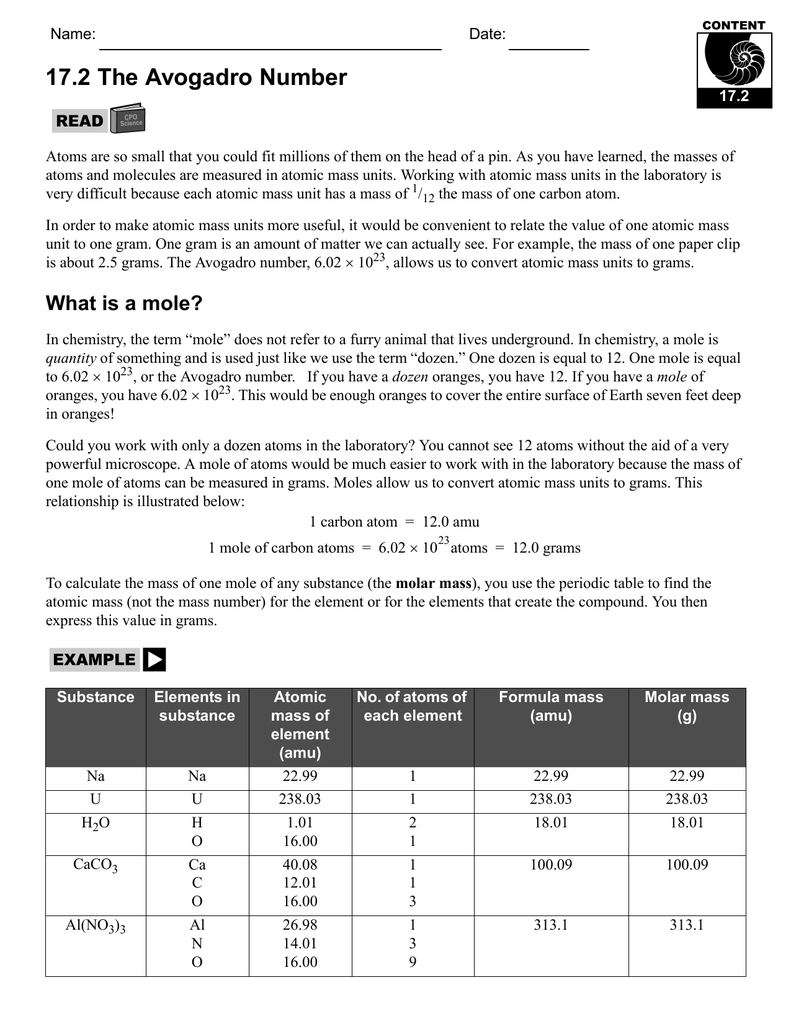
Mole Concept- Formula, Explanations, Examples, Related ... Mole Concept- A mole is defined as the amount of a substance that contains exactly the Avogadro number of ‘elementary entities’ of the given substance. The Avogadro number is represented by NA. The Mole Concept is a Convenient Method of Expressing the Amount of a Substance. To Learn more about the Mole Concept with Formulae and Examples with Videos … 5.4: Molar Mass- Mole-to-Mass and ... - Chemistry LibreTexts 23.09.2021 · Using the mole-mass calculation sequence, we can determine the required mass of SO 3 in two steps. First, we construct the appropriate molar ratio, determined from the balanced chemical equation, to calculate the number of moles of SO 3 needed. CHEMISTRY WORKSHEET # 2 MOLE PROBLEMS—THE MOLE AS … CHEMISTRY WORKSHEET # 3 AVOGADRO’S NUMBER. One important property of a mole is that it means a definite number of particles just like a dozen means a number of particles. While a dozen is only 12 particles a . mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms.
The mole and avogadro's number worksheet. How big is a mole? (Not the animal, the other one ... - TED-Ed The word “mole” suggests a small, furry burrowing animal to many. But in this lesson, we look at the concept of the mole in chemistry. Learn the incredible magnitude of the mole--and how something so big can help us calculate the tiniest particles in the world. Molar Mass | Boundless Chemistry - Lumen Learning One mole (abbreviated mol) is equal to 6.022×10 23 molecular entities (Avogadro’s number), and each element has a different molar mass depending on the weight of 6.022×10 23 of its atoms (1 mole). The molar mass of any element can be determined by finding the atomic mass of the element on the periodic table. For example, if the atomic mass of sulfer (S) is 32.066 … The Mole, Avogadro's Number, Molar Mass Avogadro's Number - The number of particles in. 7 mole of any substance. ... Molar Mass – The mass in grams of I mole of ... Answers to worksheet:.8 pages Quiz & Worksheet - Atomic Number and Mass Number | … About This Quiz & Worksheet. There's quite a lot to be learned about atoms. This article and quiz focuses on just some of that, by asking you to recall facts …
Worksheet #13 Using Avogadro's number and Molar Masses ... Using the conversion factor [ Molar Mass = 1 Mole] solve the following problems using dimensional analysis. Keep track of your units! a) Calculate the mass of ...2 pages Chemistry Simulations - CK-12 Foundation Avogadro's Number. Conversions Between Mass and Number of Particles. Conversions Between Moles and Atoms. Conversions Between Moles and Mass. Molar Mass. Mole Road Map. Chemical Equations and Reactions. Balancing Equations. Decomposition Reactions. Matter and Its States . Boiling. Changes of State. Heating and Cooling Curves. Kinetic Molecular … the mole and Avogadros number worksheet KEY.pdf - School ... One mole of a substance contains Avogadro's Number (6.02 x 1023) of molecules. How many molecules are in the quantities below? 1. 2.0 moles.1 page Mole-to-Mole Ratios and Calculations of a Chemical ... 17.11.2021 · Just to review for a moment, a mole isn't an animal. Well, it is, but not in chemistry. In chemistry, a mole is a unit of measurement, such that one mole of a …
CHEMISTRY WORKSHEET # 2 MOLE PROBLEMS—THE MOLE AS … CHEMISTRY WORKSHEET # 3 AVOGADRO’S NUMBER. One important property of a mole is that it means a definite number of particles just like a dozen means a number of particles. While a dozen is only 12 particles a . mole is a much larger number—6.02 x 1023 particles. Elements generally exist as the particles we call atoms. 5.4: Molar Mass- Mole-to-Mass and ... - Chemistry LibreTexts 23.09.2021 · Using the mole-mass calculation sequence, we can determine the required mass of SO 3 in two steps. First, we construct the appropriate molar ratio, determined from the balanced chemical equation, to calculate the number of moles of SO 3 needed. Mole Concept- Formula, Explanations, Examples, Related ... Mole Concept- A mole is defined as the amount of a substance that contains exactly the Avogadro number of ‘elementary entities’ of the given substance. The Avogadro number is represented by NA. The Mole Concept is a Convenient Method of Expressing the Amount of a Substance. To Learn more about the Mole Concept with Formulae and Examples with Videos …































0 Response to "43 the mole and avogadro's number worksheet"
Post a Comment