38 5.3 physics and the quantum mechanical model worksheet answers
CK-12 Chemistry Worksheet Name _____ Class _____ Date _____ Answer each of the questions below to show your achievement of the lesson objectives. Lesson Objective: Define chemistry. 1.Chemistry is the study of a.living systems b.the stars and planets c.all matter d.reactions in a test tube Calendar - Weebly 4 worksheets - see Google classroom - Due Wed 9-14 Per 1,2,4,6 Thurs 9-15 - Per 3 ... Physics and the Quantum Mechanical Model Lab - Locating the 1s Electron Resouces Class Notes - Chapter 5.1 ... Practice Balancing Equations Worksheet Answers Homework Balancing Chemical Equations WS #1 - Due ...
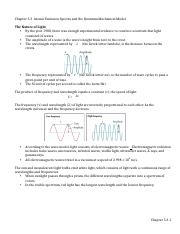
5.3 Atomic Emission Spectra And The Quantum Mechanical Model - Quizlet The Amplitude Of a Wave The waves height from zero to the crest The Wavelength Is the distance between the crests Frequency The number of wave cycles to pass a given point per unit of time - usually in cycles per second Hertz The SI unit of cycles per second - can also be expressed as a reciprocal second (2^-1) The wavelength and frequency are

5.3 physics and the quantum mechanical model worksheet answers
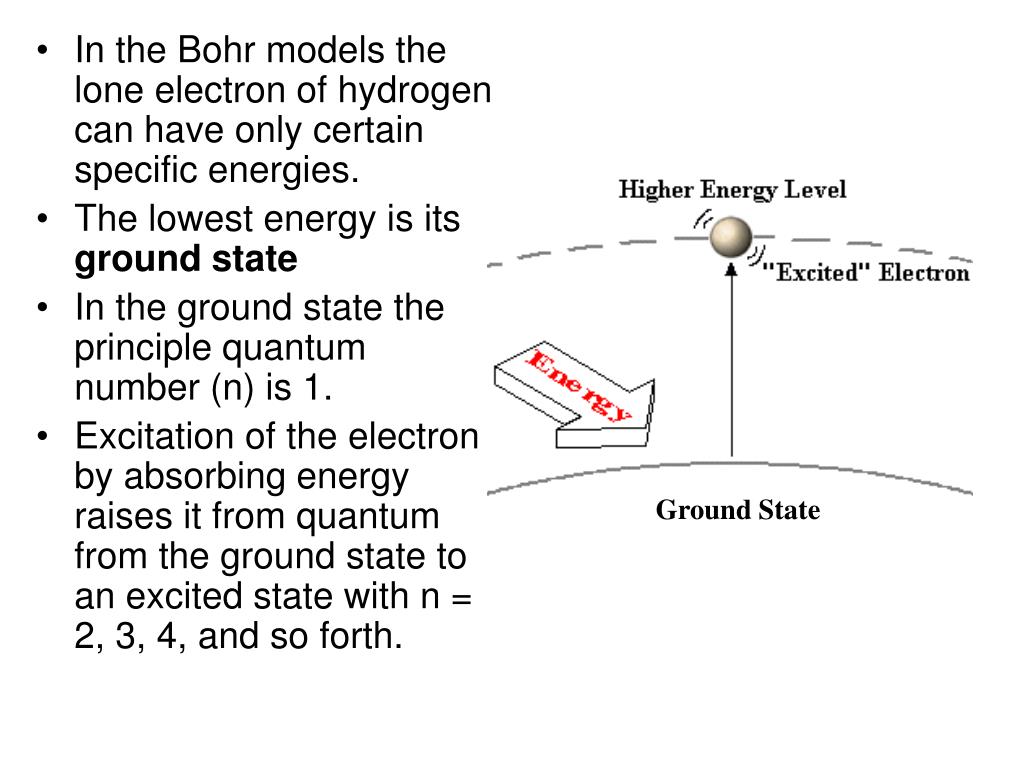
Chemistry Level 1 5.3 Physics and the Quantum Mechanical Model Light and Atomic Spectra. UNIT 3: CHEMICAL PERIODICITY Chapter 6: The Periodic Table Power Point Notes. 6.1 Organizing the Elements 6.2 Classifying the Elements 6.3 Periodic Trends Periodic Trends in Atomic Size . Chapter 7: Ionic and MetallicBonds. 7.1 Ions Appendix A: Elements Handbook HC Ch5 S3W1 - Name Date Class PHYSICS AND THE QUANTUM MECHANICAL MODEL ... Calculate the energy Calculate the wavelength Q&A For the electronic transition from n = 3 to n = 5 in the hydrogen atom. a) Calculatethe energy. b) Calculate the wavelength (in nm). Q&A The number of rescue calls received by a rescue squad in a city follows a Poisson distribution with an average of 2.83 rescues every eight hours. Heisenberg uncertainty principle (video) | Khan Academy So we take the mass of an electron, 9.11 times 10 to the negative 31st and we multiply that by the velocity, 2.2 times 10 to the sixth, and we know there's a 10% uncertainty associated with the velocity, so we get an uncertainty in the momentum 2.0 times 10 to the negative 25. So the uncertainty in the momentum is 2.0 times 10 to the negative 25.
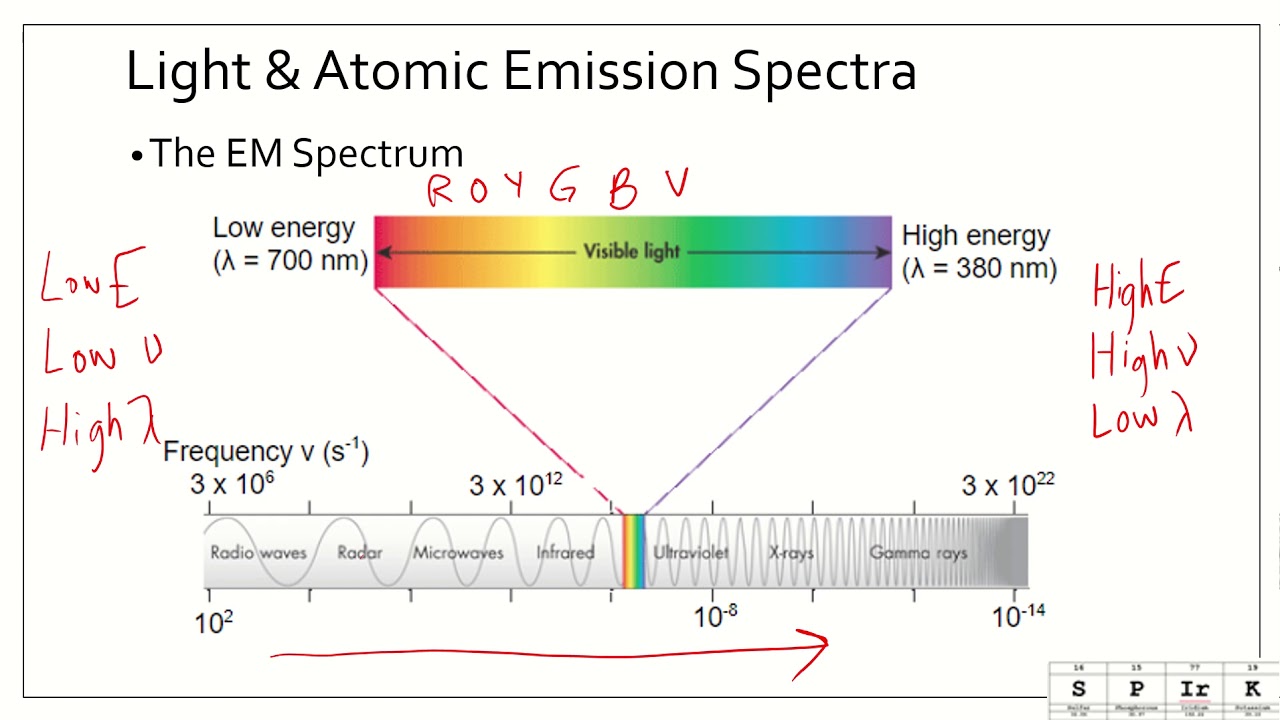
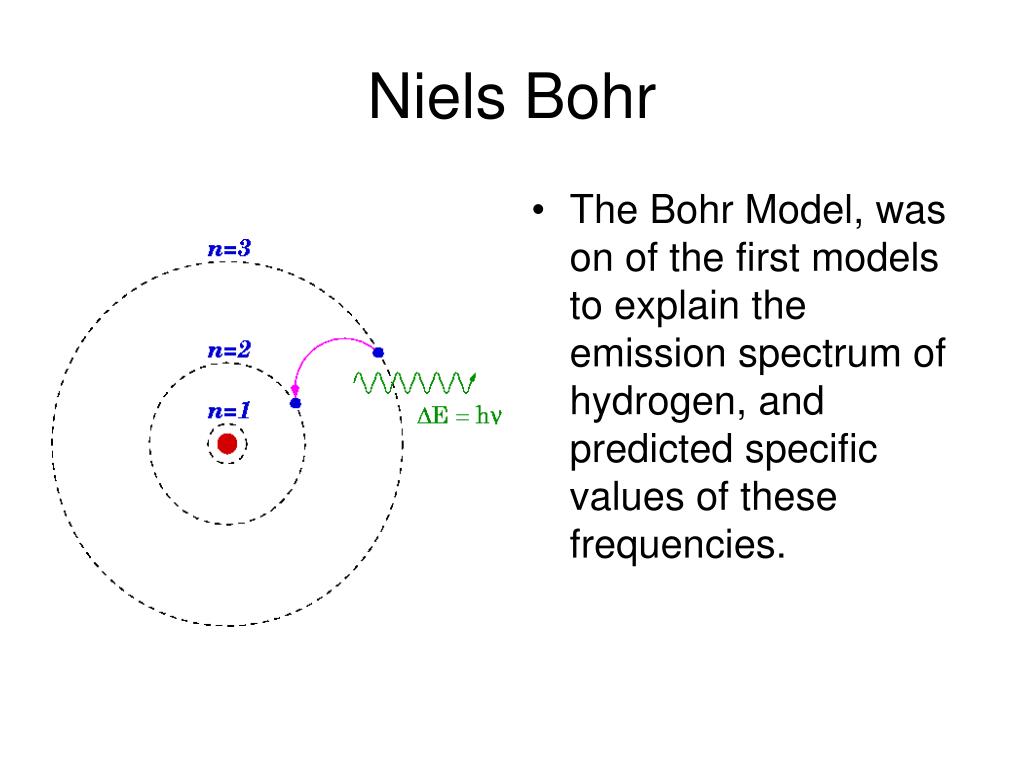
5.3 physics and the quantum mechanical model worksheet answers. Study documents, essay examples, research papers, course notes and ... Answer Key of worksheet (History of an Atom) History of Atomic Theories Worksheet Answers. Section 4.1 Studying Atoms. ... 5.3 Physics and the Quantum Mechanical Model. Isotopes and Making Atoms Hydrogen 1 Electrons in Ions Adding. Chapter 17 Resource: Properties of Atoms and the Periodic Table. 5.2 Flashcards | Quizlet The orbitals of a principle energy level are lower in energy than the orbitals in the next higher principle energy level. ST the configuration 3d2 4s2 is more stable than the configuration 3d5 4s1. NT As many as 4 e-s can occupy the same orbital. NT The pauli exclusion principle states that an atomic orbital may, at most, describe two electrons. AT 5.3 Review - Name Date Class PHYSICS AND THE QUANTUM MECHANICAL MODEL ... name date class physics and the quantum mechanical model section review objectives describe the relationship between the wavelength and frequency of light explain how the frequencies of light are related to changes in electron energies distinguish between quantum mechanics and classical mechanics identify the cause of the atomic emission spectrum … 5.3 notes - Chapter 5.3 Atomic Emission Spectra and the Quantum ... • In the visible spectrum, red light has the longest wavelength and the lowest frequency. Chapter 5.3-1 The electromagnetic spectrum consists of radiation over a broad range of wavelengths. When atoms absorb energy, their electrons move to higher energy levels. These electrons lose energy by emitting light when they return to lower energy levels.
Physics - Google Docs 3231.Math.12 Solve mechanics problems using the quadratic formula. 3231.Math.13 Find the derivative (velocity function) of a distance (displacement) function. 3231.Math.14 Find the derivative... College Physics: A Strategic Approach / Edition 4 - Barnes & Noble For courses in algebra-based introductory physics. Make physics relevant for today's mixed-majors students. College Physics: A Strategic Approach, 4th Edition expands its focus from how mixed majors students learn physics to focusing on why these students learn physics. The authors apply the best results from educational research and Mastering ™ Physics metadata to present basic physics in ... 5.3 - Atomic Emission Spectra & Quantum Mechanism Model - Quizlet 2.998 x 10^8 m/s What is spectrum? Wavelengths of visible light that are separated when a beam of light passes through a prism; range of wavelengths of electromagnetic radiation. When atoms adsorb energy, their electrons move to ______ energy levels. Higher These electrons lose energy by ________ _______ when they return to _______ energy levels. Quantum Mechanics Questions and Answers | Homework.Study.com (a) The energy and position of an electron cannot be determined simultaneously. (b) Lower energy orbitals are filled with elec... View Answer Which of the following is a reasonable criticism of the...
Section 5.3 Review.doc - Name Date Class PHYSICS AND THE QUANTUM ... PDF Particle Physics - University of Cambridge 1.1 Quantum Fields 11 1.2 Natural Units 15 A Interlude: The Road to Discovery 20 A.1 Ray Physics 20 A.2 The Electron 23 A.3 The Proton 25 A.4 The Neutron 27 2 A First Look at Quantum Fields 30 2.1 Matter Fields and Force Fields 30 2.1.1 Spin 31 2.1.2 The Dirac Equation 32 2.1.3 Anti-Matter 33 2.1.4 Massless Particles 37 2.2 Quantum ... CK-12 Physics - Intermediate Contents 16.4 Dielectrics. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 167 16.5 Electrical Energy Storage ... DOCX Honors Chemistry - Central Bucks School District Models of the Atom & Physics and the Quantum Mechanical Model. Can you… Tell me the big problem with Rutherford's model of the atom? Tell me the big question it didn't answer? Tell me who came up with a solution to this problem and how? Tell me whether an electron has higher or lower energy based on its distance from the nucleus?
interpreting ph worksheet answers 28 5.3 Physics And The Quantum Mechanical Model Worksheet Answers nuviab6ae4.blogspot.com quantum PH Problems WS Key - PH Problems Worksheet Answers 1 ANSWER E Reasoning ph problems answers ws key worksheet answer Climograph Worksheet - Wiildcreative talktoak.com
5.3 Physics and the Quantum Mechanical Model - Studyres Physics other → Top subcategories Anthropology Law Political Science Psychology Sociology ... Section 5.3 Physics and Quantum Mechanical Model. Unit 2 Intro Worksheet - Coral Gables Senior High. Slide 1. Unit 2 - Renton School District. HW 8.
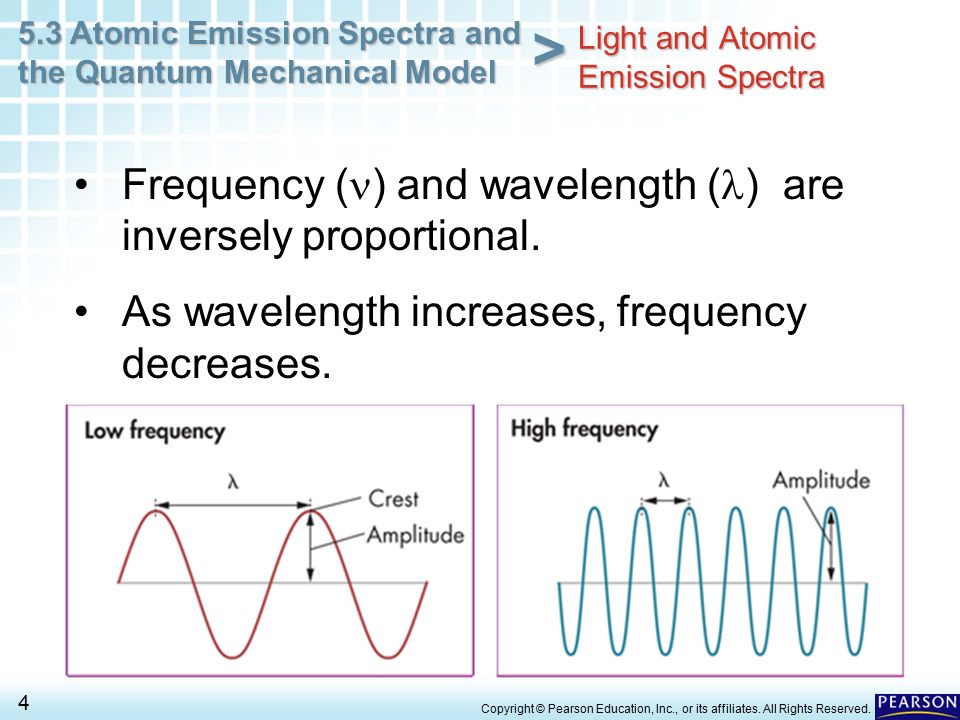
PDF chem TE ch05 - Henry County Schools Section 5.3 Physics and the Quantum Mechanical Model139 The product of frequency and wavelength always equals a constant (c), the speed of light: cλν The wavelength and frequency of light are inversely proportional to each other. As the wavelength of light increases, for example, the frequency decreases.
Worksheet_4_Answers.pdf - Chem 1A: Discussion Worksheet 4... View Worksheet_4_Answers.pdf from CHEM 1A at University of California, Irvine. Chem 1A: Discussion Worksheet 4 Answer Key 1. Explain the difference between an orbit and an orbital. An orbit
Science Chem/Physics - Mr. O'Leary / Regents Physics Science Chem/Physics - Mr. O'Leary; Science Earth & Chemistry - Mr. LeBaron; Science Living Enviro - Mrs. Miller ... Quantum Theory part II Lecture Notes ... Standard Model and Collisions worksheet . Unit Jeopardy review - reserved for class use. HW Assignments. Reading Assignments (RA) HW #1: Finish Duality and Models of the atom worksheet. HW ...
PDF 1000 Solved Problems in Modern Physics Quantum Mechanics, L.I. Schiff, 1955, to Cambridge University Press for using some valuable information from Introduction to High Energy Physics, D.H. Perkins and to Ginn and Co: and Pearson and Co: for access to Differential and Integral Calculus, William A. Granville, 1911. My thanks are due to Springer-Verlag, in
5.3 Projectile Motion - Physics | OpenStax Figure 5.29 (a) We analyze two-dimensional projectile motion by breaking it into two independent one-dimensional motions along the vertical and horizontal axes. (b) The horizontal motion is simple, because a x = 0 and v x is thus constant.
Circular Motion and Gravitation Review - Physics Classroom The object experiences a force which has a component directed parallel to the direction of motion. Inertia causes objects to move in a circle. There can be a force pushing outwards on the object as long as the net force in inwards. Because the speed is constant, the acceleration is zero. The acceleration and the net force vector are directed ...
Carter Cape - 5.3 Atomic Emission Spectra and the Quantum Mechanical Model Cell structure and function worksheet with diagrams to annotate. Practise quiz 1 first unit end of unit quiz and answers. Chapter 1 Understanding life-span human development. 04- Vocabulary-1 October 20, 20021111111. Chapter 8 Notes Entrepreneurship. SCS-100-R2491 Perspectives in the Social Sci 21EW2. homework assignment that i felt nice enough ...
5.3: Physics and the Quantum Mechanical Model - Quizlet According to quantum mechanics, the motion of subatomic particles may be described as. Wave-like. The frequency and wavelength of all wars are ___ related. Immensely. Every element emits ___ if it is heated by passing an electric discharge through its gas or vapor. Light. Passing this emission through a prism gives the ___ of the element.
5.3 Physics and the Quantum Mechanical Model - Quizlet 5.3 Physics and the Quantum Mechanical Model Flashcards Learn Test Match Flashcards Learn Test Match Created by jblake16 Terms in this set (11) Amplitude The waves height from zero to crest Wavelength The distance between the crests Frequency The number of wave cycles Hertz SI unite of cycles per second Electromagnetic Spectrum
Heisenberg uncertainty principle (video) | Khan Academy So we take the mass of an electron, 9.11 times 10 to the negative 31st and we multiply that by the velocity, 2.2 times 10 to the sixth, and we know there's a 10% uncertainty associated with the velocity, so we get an uncertainty in the momentum 2.0 times 10 to the negative 25. So the uncertainty in the momentum is 2.0 times 10 to the negative 25.
HC Ch5 S3W1 - Name Date Class PHYSICS AND THE QUANTUM MECHANICAL MODEL ... Calculate the energy Calculate the wavelength Q&A For the electronic transition from n = 3 to n = 5 in the hydrogen atom. a) Calculatethe energy. b) Calculate the wavelength (in nm). Q&A The number of rescue calls received by a rescue squad in a city follows a Poisson distribution with an average of 2.83 rescues every eight hours.
Chemistry Level 1 5.3 Physics and the Quantum Mechanical Model Light and Atomic Spectra. UNIT 3: CHEMICAL PERIODICITY Chapter 6: The Periodic Table Power Point Notes. 6.1 Organizing the Elements 6.2 Classifying the Elements 6.3 Periodic Trends Periodic Trends in Atomic Size . Chapter 7: Ionic and MetallicBonds. 7.1 Ions Appendix A: Elements Handbook

























0 Response to "38 5.3 physics and the quantum mechanical model worksheet answers"
Post a Comment