42 isotope practice worksheet answers
achieverpapers.comAchiever Papers - We help students improve their academic ... Professional academic writers. Our global writing staff includes experienced ENL & ESL academic writers in a variety of disciplines. This lets us find the most appropriate writer for any type of assignment. › createJoin LiveJournal Password requirements: 6 to 30 characters long; ASCII characters only (characters found on a standard US keyboard); must contain at least 4 different symbols;
› cms › libAtomic Structure Worksheet - Washoe County School District Give the isotope symbol and number of neutrons in one atom of the following elements. Show your calculations. Barium – 138 Sulfur – 32 Carbon – 12 Hydrogen – 1 Fluorine – 19 Magnesium – 24 Silicon - 28 Mercury – 202 Name the element which has the following numbers of particles. Be specific.

Isotope practice worksheet answers
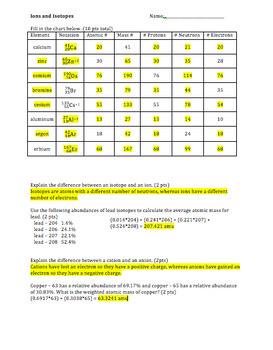
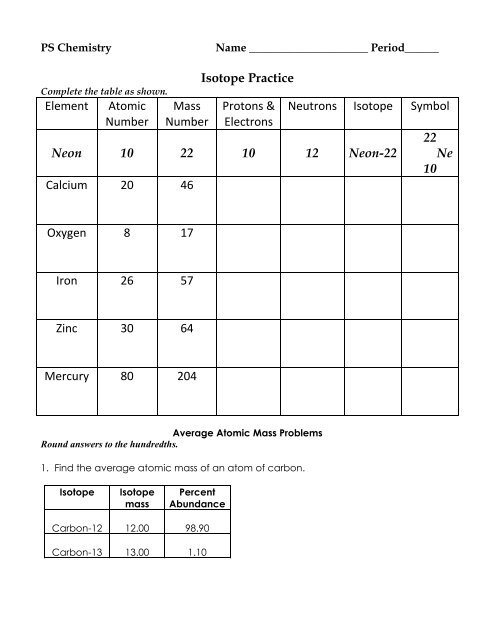
› relative-atomic-massIsotopes & Relative Atomic Mass (solutions, examples, videos) is an isotope of chlorine that has 17 protons and 18 neutrons. is an isotope of chlorine that has 17 protons and 20 neutrons. Generally, isotopes behave the same way during chemical reactions. The extra neutrons just change the mass of the atom and its density. › cms › libIsotope Worksheet Answer Key - ISD 622 Isotope Practice Worksheet Name: 1. 2. D. 4. 13 12 Here are three isotopes of an element: a. The element is: b. The number 6 refers to the c. The numbers 12, 13, and 14 refer to the d. How many protons and neutrons are in the first isotope? (O e. How many protons and neutrons are in the second isotope? f. › cms › lib6Basic Atomic Structure Worksheet Key - Neshaminy School District isotopes, taking into account the Of each isotope. The q of an element is the total number of protons and neutrons in the of atom. The mass number is used to calculate the number of O in one atom of an element. In order to calculate the number of neutrons you must subtract the from the mqSS Give the symbol of and the number of protons in one ...
Isotope practice worksheet answers. › userfiles › 2856Average Atomic Mass Practice Problems - scott.kyschools.us 11. Calculate the percentages of each isotope present in a mixture of: a) 113 In and 115 In has an average mass of 114.8 u. b) Naturally occurring silicon consists of three isotopes, 28 Si, 29 Si, and 30 Si, whose atomic masses are 27.9769, 28.9865, and 29.9838, respectively. The most abundant isotope is 28 › cms › lib6Basic Atomic Structure Worksheet Key - Neshaminy School District isotopes, taking into account the Of each isotope. The q of an element is the total number of protons and neutrons in the of atom. The mass number is used to calculate the number of O in one atom of an element. In order to calculate the number of neutrons you must subtract the from the mqSS Give the symbol of and the number of protons in one ... › cms › libIsotope Worksheet Answer Key - ISD 622 Isotope Practice Worksheet Name: 1. 2. D. 4. 13 12 Here are three isotopes of an element: a. The element is: b. The number 6 refers to the c. The numbers 12, 13, and 14 refer to the d. How many protons and neutrons are in the first isotope? (O e. How many protons and neutrons are in the second isotope? f. › relative-atomic-massIsotopes & Relative Atomic Mass (solutions, examples, videos) is an isotope of chlorine that has 17 protons and 18 neutrons. is an isotope of chlorine that has 17 protons and 20 neutrons. Generally, isotopes behave the same way during chemical reactions. The extra neutrons just change the mass of the atom and its density.

































0 Response to "42 isotope practice worksheet answers"
Post a Comment